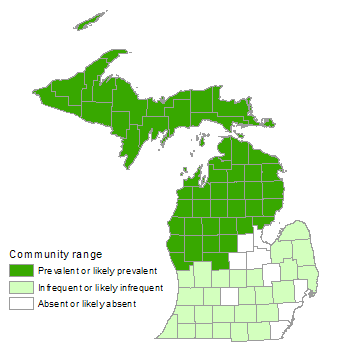
Poor Conifer Swamp
Overview
Poor conifer swamp is a nutrient-poor, forested peatland characterized by acidic, saturated peat, and the prevalence of coniferous trees, sphagnum mosses, and ericaceous shrubs. This system is found predominantly north of the climatic tension zone, and much less frequently in southern Lower Michigan. The community occurs in depressions in glacial outwash and sandy glacial lakeplains and in kettles on pitted outwash and depressions on moraines. Fire occurs naturally during drought periods and creates even-aged, often monospecific, stands of black spruce (Picea mariana). Windthrow, beaver flooding, and insect defoliation are also important disturbance factors influencing species composition and structure.

Rank
Global Rank: G4 - Apparently secure
State Rank: S4 - Apparently secure
Landscape Context
Poor conifer swamps occur in depressions on pitted outwash and moraines and on flat areas or shallow depressions in glacial outwash and glacial lakeplains. Peatlands occurring on former glacial lakebeds and drainageways tend to be more extensive than kettle peatlands, which are limited in area by the size of the glacial ice-block that formed the basin. Poor conifer swamps within large wetland complexes typically occur adjacent to other peatland communities, often grading into bog, poor fen, muskeg, patterned fen, and/or rich conifer swamp. More minerotrophic systems such as northern fen, prairie fen, shrub thicket, wet meadow, rich conifer swamp, hardwood-conifer swamp, southern hardwood swamp, and rich tamarack swamp can occur along the outer margins of poor conifer swamps where groundwater seepage from the adjacent uplands is prevalent. Poor conifer swamps often occur adjacent to lakes and meandering streams (usually less than third order). Poor conifer swamp can also occur in the first bottom of floodplain valleys in backswamps where over-the-bank flooding from the river is infrequent and organic matter can accumulate. A wide array of upland community types can occur adjacent to poor conifer swamp; some of the more frequent neighboring upland systems include dry northern forest, dry-mesic northern forest, and mesic northern forest.
Soils
The organic soils of poor conifer swamps are composed of peat, which forms a continuous mat that can be as little as 15 cm (6 in) deep but is often at least 40 cm (16 in) deep. The rooting zone within poor conifer swamps is typically quite shallow, confined to the upper 15 cm (6 in) of the surface peat. Depth of peat and soil moisture vary within a site. Peat depth is typically greatest near the center of a peatland and decreases toward the peatland margin or in areas with groundwater influence. The surface peats of poor conifer swamps are dominated by saturated fibric peat, which is loosely compacted and spongy, contains partially decomposed sphagnum moss with fragments of wood and occasionally sedge, and like the surface water, is extremely acidic, cool, and characterized by low nutrient availability and oxygen levels.
Natural Processes
Poor conifer swamps are poorly drained ombrotrophic to weakly minerotrophic peatlands, receiving inputs of water and nutrients primarily from ion-poor precipitation. Saturated and inundated conditions inhibit organic matter decomposition and allow for the accumulation of peat. Under cool, anaerobic, and acidic conditions, the rate of organic matter accumulation exceeds organic decay. Once sphagnum mosses become established on the peat mat, they maintain and enhance saturated, acidic, and cool conditions, which in turn promote continued peat development. Development and expansion of peatlands occur via two distinct processes: lake-filling and paludification. Lake-filling occurs in small lakes with minimal wave action, where gradual peat accumulation results in the development of a peat mat that can fill the basin or occur as a floating mat or grounded mat. Succession in lake-filled peatlands typically proceeds from lake to marsh to fen to bog to poor conifer swamp. Paludification is the blanketing of terrestrial systems (often forests) by the overgrowth of peatland vegetation. Paludified peatlands typically develop on flat areas (typically lakeplain) where peat builds vertically and spreads horizontally. The lateral expansion of peatland into forested systems can result in an increase in the water table and acidity and subsequent decreases in soil temperatures, nutrient availability, decomposition rates, canopy cover, growth rates, and seedling establishment. Paludification also results in a shift in species composition, with swamp conifers, especially black spruce, becoming more prevalent. For both lake-filling and paludification, peat accumulates above the water table, isolating the peatland from groundwater influence.
Disturbance factors influencing poor conifer swamps include fire, flooding, windthrow, and insects. Fire, which is an infrequent but important disturbance factor within poor conifer swamps, controls tree population dynamics by initiating and terminating succession. Estimates of fire return intervals for forested peatlands range widely from over a hundred to several hundred years in fire-prone landscapes to several hundred to over a thousand years in fire-protected landscapes. Fire severity and frequency in poor conifer swamps are closely related to climatic change and fluctuations in water level. Prolonged periods of drought and a lowered water table can allow the surface peat to dry out and burn. When the surface peat burns, the fire releases organic matter from the peat, stimulates decay, slows peat accumulation, and exposes mineral soil. Fires within poor conifer swamps are typically stand-replacing crown fires that kill the majority of canopy trees. Fires in poor conifer swamps generate a flush of growth, first in ground layer taxa and ericaceous shrubs, and then in coniferous trees. Exposure of the mineral soil provides a suitable medium for the establishment and germination of swamp conifers.
Beaver, through their dam-building activities, can instigate substantial hydrologic change to peatland systems, either causing flooding or lowering the water table of poor conifer swamps depending on the location of the forest in relation to the dam. Prolonged flooding of poor conifer swamps can result in the death of canopy trees and the conversion of forested peatlands to bogs or even open systems dominated by marsh or fen vegetation. Roots of peatland trees are physiologically active near the surface and are quickly killed when the water table rises following flooding. Flooding typically causes tree mortality after approximately ten years but different tree species have different tolerances for flooding. Short periods of flooding can cause needle chlorosis, necrotic needle tips, and decreased shoot and root growth of swamp conifers due to low oxygen concentration and nutrient availability in the water-logged rooting medium. The lowering of the water table through beaver damming or climatic changes can also significantly affect the species composition and successional trajectory of poor conifer swamps. Lowering of a forested peatland’s water table results in increased soil aeration, soil temperature, decomposition, nutrient availability, and consequently, increased tree growth. Lowered water tables can dramatically reduce the micro-scale heterogeneity that characterizes peatlands by eliminating the fine-scale gradients in pH, moisture, and nutrient availability associated with hummocks and hollows. In addition, long-term water table drawdown can cause the decomposition of the organic layer and the conversion of poor conifer swamp to a more minerotrophic forest type.
Trees growing in poor conifer swamps are particularly susceptible to windthrow because sphagnum peat provides a poor substrate for anchoring trees. The living roots of woody peatland plants occur in a shallow rooting zone, generally restricted to the uppermost few centimeters where there is sufficient oxygen to maintain aerobic respiration. The superficial rooting of trees and poor drainage result in numerous windthrows. Small-scale wind disturbance, along with insect herbivory, contributes to the structural diversity of poor conifer swamps, generating numerous snags, coarse woody debris, and gaps within the canopy. Poor conifer swamps, which often contain dense monospecific stands of trees, are inherently susceptible to epizootic attacks of insects and parasites. The plant parasite dwarf mistletoe (Arceuthobium pusillum) can increase the mortality of black spruce. Three insect defoliators are prevalent in peatlands: larch sawfly (Pristiphora erichsonii), larch casebearer (Coleophora laricella), and spruce budworm (Choristoneura fumiferana). Spruce budworm defoliates both black spruce and balsam fir but tends to be more detrimental to the latter. Tamarack growing in poor conifer swamps often suffers from repeated defoliation by larch sawfly. Although a more recent arrival in Michigan, the larch casebearer is beginning to cause heavy defoliation to tamarack, especially in the eastern and central Upper Peninsula.
Vegetation
Poor conifer swamps are characterized by a canopy of coniferous trees, low ericaceous, evergreen shrubs, a poor herbaceous layer, and a hummocky carpet of sphagnum moss. The canopy is often dominated by black spruce. Tamarack (Larix laricina) is also a frequent canopy dominant or codominant. Canopy associates include balsam fir (Abies balsamea), white pine (Pinus strobus), jack pine (Pinus banksiana), paper birch (Betula papyrifera), and American mountain ash (Sorbus americana). The shrub layer is dominated by low, ericaceous shrubs, with Labrador tea (Ledum groenlandicum) being the most prevalent. Additional heath shrubs include bog rosemary (Andromeda glaucophylla), leatherleaf (Chamaedaphne calyculata), creeping snowberry (Gaultheria hispidula), wintergreen (G. procumbens), huckleberry (Gaylussacia baccata), sheep-laurel (Kalmia angustifolia), bog laurel (K. polifolia), low sweet blueberry (Vaccinium angustifolium), Canada blueberry (V. myrtilloides), and small cranberry (V. oxycoccos). The tall shrub layer is less dense than the low shrub layer and is often restricted to the periphery of the swamp or adjacent to streams that may meander through the peatland. Tall shrubs include black chokeberry (Aronia prunifolia), wild-raisin (Viburnum cassinoides), and mountain holly (Nemopanthus mucronata). Stunted trees, especially black spruce and tamarack, may also occur within the understory. The herbaceous layer is species depauperate. Several characteristic sedges include wiregrass sedge (Carex lasiocarpa), bristly-stalked sedge (C. leptalea), few-flower sedge (C. pauciflora), few-seed sedge (C. oligosperma), and three-seeded sedge (C. trisperma), with the last two being most common. Additional graminoids include sheathed cotton-grass (Eriophorum spissum), tawny cotton-grass (E. virginicum), and wool-grass (Scirpus cyperinus). Additional prevalent ground flora include bluebead lily (Clintonia borealis), goldthread (Coptis trifolia), bunchberry (Cornus canadensis), fireweed (Epilobium angustifolium), wild blue flag (Iris versicolor), northern bugleweed (Lycopus uniflorus), Canada mayflower (Maianthemum canadense), false mayflower (Smilacina trifolia), and starflower (Trientalis borealis). Characteristic ferns include sensitive fern (Onoclea sensibilis), royal fern (Osmunda regalis), and Virginia chain-fern (Woodwardia virginica). Insectivorous plants, such as round-leaved sundew (Drosera rotundifolia) and pitcher-plant (Sarracenia purpurea), are occasional features of poor conifer swamps. The continuous moss layer of poor conifer swamps is typically dominated by sphagnum mosses, especially Sphagnum angustifolium, S. capillifolium, S. fuscum, S. magellanicum, and S. recurvum. Additional mosses can include S. capillaceum, S. centrale, S. compactum, S. cuspidatum, S. papillosum, S. wulfianum, and Drepanocladus aduncus. Shade beneath black spruce trees is conducive to the growth of feather moss (Pleurozium schreberi). Plant species diversity within poor conifer swamps is strongly correlated to the hummock-hollow microtopography; each individual hummock in a peatland is in essence a miniature ecosystem with distinct gradients in water and substrate chemistry, soil moisture, aeration, and nutrients.
For information about plant species, visit the Michigan Flora website.
Plant Lists
Graminoids
- sedges (Carex oligosperma, C. pauciflora, C. trisperma, and others)
- sheathed cotton-grass (Eriophorum vaginatum)
- tawny cotton-grass (Eriophorum virginicum)
- wool-grass (Scirpus cyperinus)
Forbs
- wild calla (Calla palustris)
- bluebead lily (Clintonia borealis)
- goldthread (Coptis trifolia)
- pink lady-slipper (Cypripedium acaule)
- round-leaved sundew (Drosera rotundifolia)
- wild blue flag (Iris versicolor)
- Canada mayflower (Maianthemum canadense)
- false mayflower (Maianthemum trifolium)
- pitcher-plant (Sarracenia purpurea)
- starflower (Trientalis borealis)
Ferns
- sensitive fern (Onoclea sensibilis)
- royal fern (Osmunda regalis)
- Virginia chain-fern (Woodwardia virginica)
Mosses
- ribbed bog moss (Aulacomnium palustre)
- big red stem moss (Pleurozium schreberi)
- pohlia moss (Pohlia nutans)
- sphagnum mosses (Sphagnum spp.)
Shrubs
- bog rosemary (Andromeda glaucophylla)
- black chokeberry (Aronia prunifolia)
- leatherleaf (Chamaedaphne calyculata)
- bunchberry (Cornus canadensis)
- creeping snowberry (Gaultheria hispidula)
- wintergreen (Gaultheria procumbens)
- huckleberry (Gaylussacia baccata)
- mountain holly (Ilex mucronata)
- winterberry (Ilex verticillata)
- sheep laurel (Kalmia angustifolia)
- bog laurel (Kalmia polifolia)
- Labrador-tea (Rhododendron groenlandicum)
- blueberries (Vaccinium angustifolium, V. corymbosum, and V. myrtilloides)
- small cranberry (Vaccinium oxycoccos)
- wild-raisin (Viburnum cassinoides)
Trees
- balsam fir (Abies balsamea)
- paper birch (Betula papyrifera)
- tamarack (Larix laricina)
- black spruce (Picea mariana)
- jack pine (Pinus banksiana)
- white pine (Pinus strobus)
Noteworthy Animals
In general, animal diversity is low in poor conifer swamps because of the low productivity of peatland plants, the unpalatability of the vegetation, and the high acidity of the peat. Selective browsing by moose (Alces alces, state threatened) can result in changes to poor conifer swamp floristic composition and structure. Beaver (Castor canadensis) can profoundly influence the hydrology of poor conifer swamp through their dam-building activities.
Rare Plants
- Carex wiegandii (Wiegand's sedge, state threatened)
- Empetrum nigrum (black crowberry, state threatened)
- Luzula parviflora (small-flowered wood rush, state threatened)
- Rubus acaulis (dwarf raspberry, state endangered)
- Sarracenia purpurea f. heterophylla (yellow pitcher-plant, state threatened)
Rare Animals
- Alces alces (moose, state threatened)
- Ardea herodias (great blue heron, protected by the Migratory Bird Treaty Act of 1918)
- Canis lupus (gray wolf, state threatened)
- Emydoidea blandingii (Blanding’s turtle, state special concern)
- Falcipennis canadensis (spruce grouse, state special concern)
- Falco columbarius (merlin, state threatened)
- Glyptemys insculpta (wood turtle, state special concern)
- Haliaeetus leucocephalus (bald eagle, state threatened)
- Lynx canadensis (lynx, state endangered)
- Pandion haliaetus (osprey, state threatened)
- Picoides arcticus (black-backed woodpecker, state special concern)
- Sistrurus c. catenatus (eastern massasauga, federal candidate species and state special concern)
- Tympanuchus phasianellus (sharp-tailed grouse, state special concern)
Biodiversity Management Considerations
When the primary conservation objective is to maintain biodiversity of poor conifer swamps, the best management strategy is to leave large tracts unharvested and allow natural processes (fire, beaver flooding, and insect defoliation) to operate unhindered and stochastically generate a range of successional stages. It is crucial to allow dead and dying wood to remain within these systems to become snags, stumps, and fallen logs. Within areas managed solely for biodiversity, resource practitioners should refrain from salvage harvesting following fire, wind, and insect disturbance. Salvage logging, especially following fire, can severely diminish nutrient pools and site productivity in addition to reducing structural heterogeneity.
Where poor conifer swamps are being actively managed, maintaining poor conifer swamps in different age-classes and stages of structural development at the landscape level will contribute to the preservation of regional biodiversity. Increasing old-growth and over-mature poor conifer swamps and their habitat characteristics can be accomplished by extending the rotation period of these systems. Long rotation periods (more than 100 years) are favorable for numerous species, such as epiphytic lichen and trunk foraging birds that depend on old, large trees. Within fire-prone landscapes, managers should consider using even-aged management, while in fire-resistant systems uneven-aged silviculture is more appropriate. Partial and selective cutting within poor conifer swamps has been suggested to simulate secondary disturbance such as windthrow. However, these uneven-aged cutting techniques do not generate pit and mound microtopography and fail to leave behind the same volume of coarse wood generated by natural disturbance such as windthrow. Even-aged management of poor conifer swamps should be restricted to fire-prone landscapes, and timber rotations should reflect site-specific fire return intervals. A common misconception about even-aged management of forested peatlands is that clear-cuts or final harvests are surrogates for crown fires. Even-aged management within conifer swamps should strive to maintain patches of residual trees and numerous snags. Scattered seed trees and cone-bearing logging slash can provide an important source for natural regeneration. With both even-aged and uneven-aged management of forested peatlands, protecting the residual regeneration and seed trees and generating or preserving suitable sites for seedling establishment are critical. “Careful logging” is stressed by numerous researchers as a means of preserving advanced regeneration. Harvesters can avoid damage to residual trees by planning ahead of time where to travel, where to drop felled trees (directional felling), and where to process and pile the bucked logs. Impacts to peat soils can be minimized by using high-flotation tires or restricting operation in forested peatlands to winter when snow cover and frozen soils provide protection from rutting. Integral to the maintenance of forested peatlands is the preservation of hydrologic regimes. The installation and maintenance of culverts under roads passing through peatlands can avert flooding and drying.
In addition to direct impacts to vegetation, alteration of peatland hydrology due to road building, creation of drainage ditches and dams, and runoff from logging and agriculture has led to the significant changes in peatland composition and structure. Flooding of poor conifer swamps can cause the death of canopy trees and the conversion of forested peatland to open wetlands. Roads and highways traversing large peatlands complexes, especially in the Upper Peninsula, have blocked drainage, causing flooding, tree mortality, and conversion to open wetlands. Conversely, ditches and drains that lower water tables can result in rapid decomposition of peat and increased shrub and tree encroachment.
Monitoring and control efforts to detect and remove invasive species are critical to the long-term viability of poor conifer swamp. Particularly aggressive invasive species that may threaten the diversity and community structure include glossy buckthorn (Rhamnus frangula), narrow-leaved cat-tail (Typha angustifolia), hybrid cat-tail (Typha xglauca), reed canary grass (Phalaris arundinacea), and reed (Phragmites australis). At present, most invasive species appear to be restricted to the margins of the poor conifer swamps, where they occur in moats or ditches along roads and trails that border the community. Monitoring and control efforts to detect and remove invasive species before they become widespread are critical to the long-term viability of poor conifer swamp.
Variation
Subtle variations in overall species composition and physiognomy of poor conifer swamp occur across its range along north-south and east-west climatic gradients. Poor conifer swamps are common throughout the northern Lower Peninsula and the Upper Peninsula and are rare south of the climatic tension zone. In the southern part of the Lower Peninsula, vegetation composition is influenced by minerotrophy and a warmer climate. Tall shrubs, particularly smooth highbush blueberry (Vaccinium corymbosum), are more prevalent in southern systems.
Similar Natural Communities
Bog, muskeg, northern fen, patterned fen, poor fen, rich tamarack swamp, and rich conifer swamp.
Places to Visit
- Nine Mile Swamp, Roscommon State Forest Management Unit, Roscommon Co.
- Prison Camp Swamp, Tahquamenon Falls State Park, Luce Co.
- Sylvania, Sylvania Wilderness and Recreation Area, Ottawa National Forest, Gogebic Co.
- Tahqua Trail Swamp, Tahquamenon Falls State Park, Chippewa Co.
- Waterloo Black Spruce Bog, Waterloo State Recreation Area, Jackson Co.
Relevant Literature
- Cohen, J.G. 2006. Natural community abstract for poor conifer swamp. Michigan Natural Features Inventory, Lansing, MI. 20 pp.
- Futyma, R.P., and N.G. Miller. 1986. Stratigraphy and genesis of the Lake Sixteen peatland, northern Michigan. Canadian Journal of Botany 64: 3008-3019.
- Gates, F.C. 1942. The bogs of northern Lower Michigan. Ecological Monographs 12(3): 213-254.
- Graham, S.A. 1956. The larch sawfly in the Lake States. Forest Science 2(2): 132-160.
- Groot, A., and B.J. Horton. 1994. Age and size structure of natural and second-growth peatland Picea mariana stands. Canadian Journal of Forest Research 24: 225-233.
- Harper, K.A., Y. Bergeron, P. Drapeau, S. Gauthier, and L. DeGrandpre. 2005. Structural development following fire in black spruce boreal forest. Forest Ecology and Management 206: 293-306.
- Heinselman, M.L. 1963. Forest sites, bog processes, and peatland types in the Glacial Lake Region, Minnesota. Ecological Monographs 33(4): 327-374.
- Islam, M.A., and S.E. MacDonald. 2004. Ecophysiological adaptations of black spruce (Picea mariana) and tamarack (Larix laricina) seedlings to flooding. Trees 18: 35-42.
- Liefers, V.J., and R.L. Rothwell. 1987. Rooting of peatland black spruce and tamarack in relation to depth of water table. Canadian Journal of Botany 65: 817-821.
- MacDonald, S.E., and F. Yin. 1999. Factors influencing size inequality in peatland black spruce and tamarack: Evidence from post-drainage release growth. Journal of Ecology 87: 404-412.
- MacDonell, M.R., and A. Groot. 1997. Harvesting peatland black spruce: Impacts on advance growth and site disturbance. Forestry Chronicle 73(2): 249-255.
- Newton, P.F., and P.A. Jolliffe. 1998. Temporal size-dependent growth responses within density-stressed black spruce stands: Competition processes and budworm effects. Forest Ecology and Management 111: 1-13.
- Newton, P.F., and V.G. Smith. 1988. Diameter distributional trends within mixed black spruce/balsam fir and pure black spruce stand types. Forest Ecology and Management 25: 123-138.
- Pepin, S., A.P. Plamondon, and A. Britel. 2002. Water relations of black spruce trees on a peatland during wet years and dry years. Wetlands 22(2): 225-233.
- Ruel, J.-C., R. Horvath, C.H. Ung, and A. Munson. 2004. Comparing height growth and biomass production of black spruce trees in logged and burned stands. Forest Ecology and Management 193: 371-384.
- Schwintzer, C.R. 1981. Vegetation and nutrient status of northern Michigan bogs and conifer swamps with a comparison to fens. Canadian Journal of Botany 59: 842-853.
- Schwintzer, C.R., and T.J. Tomberlin. 1982. Chemical and physical characteristics of shallow ground waters in northern Michigan bogs, swamps, and fens. American Journal of Botany 69(8): 1231-1239.
- Stanek, W. 1961. Natural layering of black spruce in northern Ontario. Forestry Chronicle 37: 245-258.
- Taylor, S.J., T.J. Carleton, and P. Adams. 1988. Understory vegetation change in a Picea mariana chronosequence. Vegetatio 73(2): 63-72.
For a full list of references used to create this description, please refer to the natural community abstract for Poor Conifer Swamp.
More Information
Citation
Cohen, J.G., M.A. Kost, B.S. Slaughter, D.A. Albert, J.M. Lincoln, A.P. Kortenhoven, C.M. Wilton, H.D. Enander, M.E. Anderson, M.R. Parr, T.J. Bassett, and K.M. Korroch. 2025. Michigan Natural Community Classification [web application]. Michigan Natural Features Inventory, Michigan State University Extension, Lansing, MI. Available https://mnfi.anr.msu.edu/communities/classification. (Accessed: May 16, 2026).
Kost, M.A., D.A. Albert, J.G. Cohen, B.S. Slaughter, R.K. Schillo, C.R. Weber, and K.A. Chapman. 2007. Natural Communities of Michigan: Classification and Description. Michigan Natural Features Inventory, Report No. 2007-21, Lansing, MI.