Plants and Animals
Potamilus alatus Pink heelsplitter
Key Characteristics
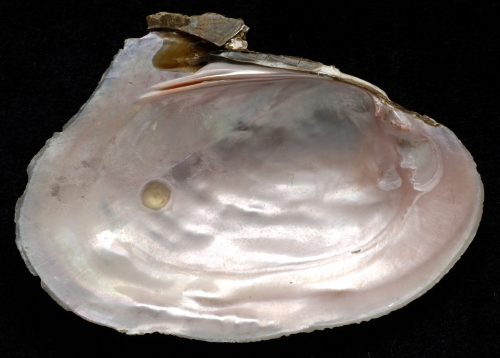
The pink heelsplitter has a large dorsal wing that extends above the hinge line. Its outline is an egg-shaped ellipse that is broad at the posterior end and narrow at the anterior end. The beak is moderately, but broadly inflated. Maximum length is 19 cm and this mussel’s life span is around 15 years. Beak sculpture consists of small variably shaped ridges that are often not visible in older individuals. The shell’s color ranges from dark brown and black to greenish tan. Green rays are visible on lighter colored individuals. The cardinal and lateral teeth are well developed, and the lateral teeth become more curved as the mussel grows older. The beak cavity is broad and shallow. The nacre color is pink, rose, or purple.
Status and Rank
US Status: No Status/Not Listed
State Status: SC - Special Concern (rare or uncertain; not legally protected)
Global Rank: G5 - Secure
State Rank: SNR - Not ranked
Occurrences
| County | Number of Occurrences | Year Last Observed |
|---|---|---|
| Allegan | 2 | 2000 |
| Bay | 2 | 2019 |
| Berrien | 2 | 1937 |
| Genesee | 1 | 2008 |
| Huron | 1 | 1931 |
| Iosco | 2 | 2003 |
| Kalamazoo | 1 | 2018 |
| Kent | 1 | 2018 |
| Macomb | 6 | 2017 |
| Midland | 2 | 2015 |
| Monroe | 13 | 2020 |
| Muskegon | 2 | 1936 |
| Ottawa | 6 | 2019 |
| Saginaw | 5 | 2011 |
| St. Clair | 16 | 2019 |
| St. Joseph | 1 | 2005 |
| Wayne | 27 | 2020 |
Information is summarized from MNFI's database of rare species and community occurrences. Data may not reflect true distribution since much of the state has not been thoroughly surveyed.
Habitat
Pink heelsplitter inhabit medium to large rivers and lakes, including Lake Erie, Lake St. Clair, and Lake Huron (Saginaw Bay). They are often found in mud, sand, and gravel substrates.
Natural Community Types
- Great lake, littoral, benthic
- Inland lake, littoral, benthic
- Mainstem stream (3rd-4th order), pool
- Mainstem stream (3rd-4th order), run
- Mainstem stream (3rd-4th order), riffle
- River (5th-6th order), pool
- River (5th-6th order), run
- River (5th-6th order), riffle
For each species, lists of natural communities were derived from review of the nearly 6,500 element occurrences in the MNFI database, in addition to herbarium label data for some taxa. In most cases, at least one specimen record exists for each listed natural community. For certain taxa, especially poorly collected or extirpated species of prairie and savanna habitats, natural community lists were derived from inferences from collection sites and habitat preferences in immediately adjacent states (particularly Indiana and Illinois). Natural communities are not listed for those species documented only from altered or ruderal habitats in Michigan, especially for taxa that occur in a variety of habitats outside of the state.
Natural communities are not listed in order of frequency of occurrence, but are rather derived from the full set of natural communities, organized by Ecological Group. In many cases, the general habitat descriptions should provide greater clarity and direction to the surveyor. In future versions of the Rare Species Explorer, we hope to incorporate natural community fidelity ranks for each taxon.
Management Recommendations
The pink heelsplitter is at risk from point source pollution, non-point source pollution, alteration of natural stream flow patterns, barriers to host fish passage, direct alteration of habitat, and invasive zebra mussels.
Limit or eliminate point source discharges of ammonia, chloride, sulfate, heavy metals, and other substances toxic to native mussels. Avoid use of aquatic herbicides containing copper whenever possible. Maintain riparian buffers along streams and promote land conservation programs to help reduce excessive erosion, sedimentation, and nutrient input from non-point sources. Maintain integrity of headwater streams and wetlands, which support the ecological function of downstream river habitats. Improve passage for fish by removing unnecessary dams and upgrading poor stream/road crossings such as culverts that are too small or are perched. Pink heelsplitter and other native mussels require fish hosts to complete their life cycle and rely on fish passage to travel to new habitats and facilitate gene flow among mussel populations. Avoid dredging, channelization, and other in-stream impacts whenever possible.
The spread of zebra mussels into Michigan’s streams and lakes remains a serious threat throughout the pink heelsplitter’s range. Pink heelsplitter populations in the Great Lakes have been reduced, and in some areas extirpated by zebra mussels. Reduce the impact of zebra mussels by avoiding the transport of water or aquatic plants – which can contain zebra mussel larvae – from one body of water to another while boating, fishing, hunting, and researching. Washing boats, trailers, and gear, and letting them dry overnight reduces the potential for spreading zebra mussels.
Active Period
Gravid from first week of May to fourth week of October
Survey Methods
In water shallower than waist deep, wading with a glass bottom bucket and, if water quality is good, swimming with a snorkel and mask are effective ways to search for pink heelsplitter. SCUBA or other dive gear is required for greater water depths. River and lake substrate should be searched both visually and tactilely, by sweeping the fingers through the substrate. Though pink heelsplitter are typically found at least partly exposed to the water column, they can be completely buried at times and difficult to detect visually. Empty shells may be found along riverbanks and occasionally in shell middens created by predators such as muskrats. A scientific collector’s permit is required to possess live native mussels shell or live individuals. Mussel surveys related to permitted projects in rivers and lakes should follow the Michigan Freshwater Mussel Survey Protocols and Relocation Procedures. Federal and/or state threatened and endangered species permits must be obtained prior to surveying for native mussels within waterbodies likely to support these species. Refer to the Mussel Map Viewer available on MNFI’s website.
Glass-bottom bucket less than waist deep water
Survey Period: From first week of June to second week of October
SCUBA greater than waist deep water
Survey Period: From first week of June to second week of October
Survey Method Comment: Surface supplied diving may be appropriate in some cases.
Snorkeling searches
Survey Period: From first week of June to second week of October
Survey Method Comment: Use this method only when water quality and conditions are safe for swimming.
References
Survey References
- Hanshue, S., J. Rathbun, P. Badra, J. Bettaso, B. Hosler, J. Pruden, and J. Grabarkiewicz. 2019. Michigan Freshwater Mussel Survey Protocols and Relocation Procedures [for rivers], Version 2. Michigan Department of Natural Resources.
- Metcalfe-Smith, J., A. MacKenzie, I. Carmichael, and D. McGoldrick. 2005. Photo Field Guide to the Freshwater Mussels of Ontario. St. Thomas Field Naturalist Club Inc, 60 pp.
- Mulcrone, R.S., and J.E. Rathbun. 2018. Field Guide to the Freshwater Mussels of Michigan. Michigan Department of Natural Resources, 59 pp.
- Watters, G. Thomas, Michael A. Hoggarth, and David H. Stansbery. 2009. The Freshwater Mussels of Ohio. The Ohio State University Press, Columbus. 421 pp.
Technical References
- Blevins, E., L. McMullen, S. Jepsen, M. Blackburn., A. Code, and S.H. Black. 2017. Conserving the Gems of Our Waters: Best Management Practices for Protecting Native Western Freshwater Mussels During Aquatic and Riparian Restoration, Construction, and Land Management Projects and Activities. 108 pp. Portland, OR: The Xerces Society for Invertebrate Conservation.
- Brim-Box, J., and J. Mossa. 1999. Sediment, land use, and freshwater mussels: Prospects and problems. Journal of the North American Benthological Society 18: 99-117.
- Colvin, S.A.R., S.M.P. Sullivan, P.D. Shirey, R.W. Colvin, K.O. Winemiller, R.M. Hughes, K.D. Fausch, D.M. Infante, J.D. Olden, K.R. Bestgen, R.J. Danehy, and L. Eby. 2019. Headwater streams and wetlands are critical for sustaining fish, fisheries, and ecosystem services. American Fisheries Society Special Report. Fisheries 44: 73-91.
- Freshwater Mollusk Conservation Society. 2016. A national strategy for the conservation of native freshwater mollusks. Freshwater Mollusk Biology and Conservation 19: 1-21.
- Haag, W.R. 2012. North American Freshwater Mussels: Natural History, Ecology, and Conservation. Cambridge University Press, New York, 505 pp.
- Strayer, D.L. and D.R. Smith. 2003. A Guide to Sampling Freshwater Mussel Populations. American Fisheries Society Monograph 8, Bethesda. 103pp.
- Watters, G.T. 1996. Small dams as barriers to freshwater mussels (Bivalvia, Unionoida) and their hosts. Biological Conservation 75: 79-85.